How can I prove this equation: "mvr" = "nh"/(2π)?
2 Answers
May 29, 2018
According to Bohr’s atomic model electron revolves around the nucleus in circular orbits.
As electron shows both wave and particle nature the circumference of circular orbit should by an integral multiple of wavelength (
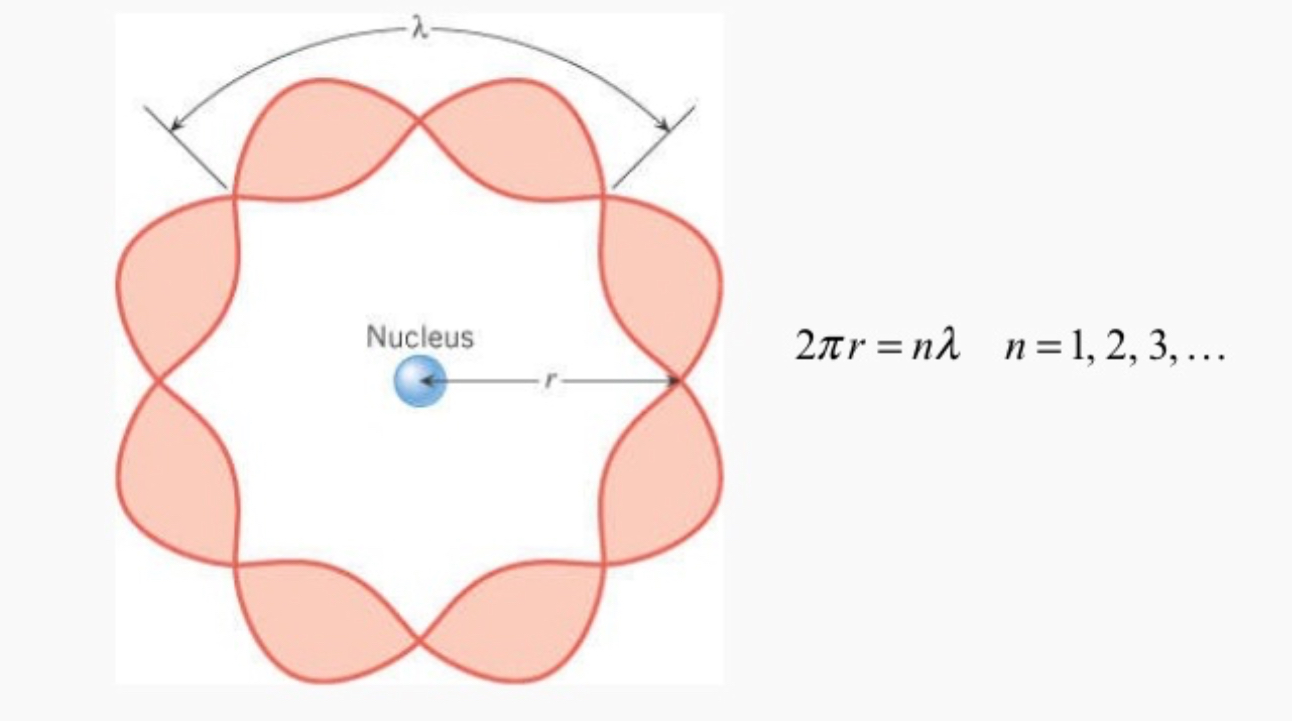
"2πr = nλ"
de-broglie wavelength (
λ = "h"/"mv"
Substitute
"2πr = n" × "h"/"mv"
On rearranging above equation
"mvr" = "nh"/"2π"
May 29, 2018
Quantisation of angular momentum is not something you prove as such. It is one of Bohr's postulates. Something that is just assumed to be true.


