How can polarity of molecules be predicted from their geometry?
1 Answer
When two identical atoms, e.g., two fluorine atoms, are connected by a covalent bond, the electrons are shared equally, and the bond is nonpolar.

However, if two different atoms are joined by a covalent bond, one atom usually has a greater tendency to attract the shared electrons toward itself. The atom with the greater electron-attracting ability has a greater electronegativity. It will have a partial negative charge, and the other atom will have a partial positive charge. We say that the bond is polar and has a dipole, because it has a partially positive “pole” and a partially negative “pole”.

We represent the dipole as an arrow with a ”plus” sign at one end pointing in the direction a positive charge would go — towards the negative end of the bond.
If a molecule has a polar bond, as in hydrogen fluoride, the centre of negative charge will be in one place (on the F atom), and the centre of positive charge will be in a different place (on the H atom). The molecule as a whole will also be polar.
If a molecule has more than one polar bond, the molecule will be polar or nonpolar, depending on how the bonds are arranged. If the polar bonds are arranged symmetrically, the bond dipoles cancel and do not create a molecular dipole. For example, the three bonds in a molecule of BF₃ are polar, but they point at angles of 120° around the central boron atom. The centre of positive charge is on the boron atom, and the centre of the three negative charges is also on the boron atom. Since the centres of positive and negative charges coincide, there is no dipole moment, and the molecule is nonpolar.

The O-H bond in a water molecule is polar, with its negative end at the O atom and its positive end at the H atom. The centre of negative charge is on the O atom, and the centre of positive charge is half-way between the two H atoms. Since the centres of positive and negative charge do not coincide, a water molecule is polar
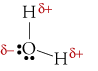
There is an excellent handout of the various shapes of molecules. It shows which ones are polar and which are nonpolar despite having polar bonds. You can find the handout at
http://www.angelo.edu/faculty/kboudrea/index/Handout_Lewis_VSEPR.pdf


