How do covalent bonds conduct electricity?
1 Answer
Sigma bonds conduct a small amount of electricity by quantum tunnelling, but delocalized pi bonds are good conductors of electricity..
Explanation:
SIGMA BONDS
Most chemists say that covalent bonds do not conduct electricity, because the electrons are trapped in the sigma bonds.
But a quantum mechanical effect called quantum tunnelling allows the electrons to tunnel through the energy barriers.
However, the effect is so small that compounds with only sigma bonds are extremely poor conductors.
For most purposes, we say that sigma bonds do not conduct electricity.

Can you spot the error in the bather's argument?
CONJUGATED π BONDS
Graphite
Graphite is a good conductor of electricity.
It consists of layered sheets of benzene rings all fused together.
http://rickwilsondmd.typepad.com/.a/6a01156e42deab970c0133f4de294b970b-pi
As in benzene, each carbon is
In each layer of graphite, the
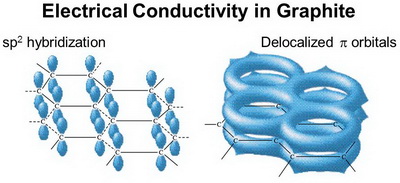
(from slideplayer.com)
Hence, graphite can conduct electricity from one side of a sheet to the other.
Graphene
Graphene is a single sheet of graphite.

As with graphite, the π electrons are delocalized and free to roam from one edge to the other.
Carbon nanotubes
Carbon nanotubes are seamless cylindrical fibres made from a single sheet of graphene.

Their electron delocalization means electrical charge can move freely along a nanotube.

