How do electron capture detectors work?
1 Answer
In general, the purpose of an electron capture detector is to detect the concentration of the analyte based on how many electrons it absorbs to use in an electron capture event.
Suppose we had a gas chromatograph that sent its output stream of carrier gas (the mobile phase) through the column labeled
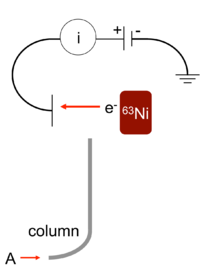 https://upload.wikimedia.org/
https://upload.wikimedia.org/
Typically, about
This emits radioactive electrons in the presence of, typically,
As the electron emitter emits electrons at the makeup gas, these electrons collide with them and ionize them, thus increasing the number of free electrons.
The free electrons travel through towards a positively-charged anode, creating a circuit. This generates the background signal. This set of electrons is then accelerated into the electron capture detector which counts electrons as it absorbs them.
It then converts the signal (like a transducer) in such a way that you get the analyte concentration.

