How do oxidation numbers relate to valence electrons?
1 Answer
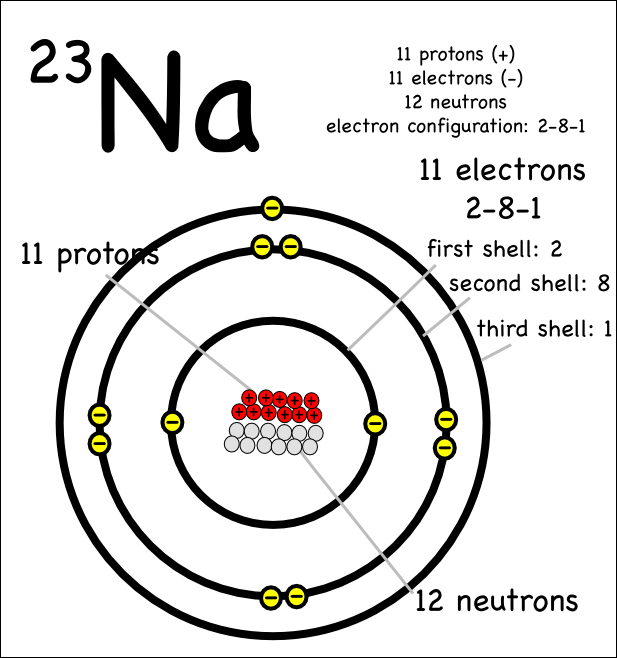
The valence electrons determine how many electrons an atom is willing to give up or how many spaces need to be filled in order to satisfy the rule of octet.
Lithium (Li), Sodium (Na) and Potassium (K) all have an electron configuration that ends as
Oxygen (O) and Sulfur (S) all have an electron configuration that ends as
There are exceptions to the rules and the transition metals usually have more than one oxidation state.
I hope this was helpful.
SMARTERTEACHER