How do you know when copper(II) sulfate hydrated water evaporates?
When considering #CuSO_4 * x H_2O# , does the number of waters, or #x# have to be a whole number? If you were to get rid of the water molecules and just have the copper sulfate, how would I know when it has evaporated?
When considering
2 Answers
The color changes!
Explanation:
To answer your first question, yes,
#"CuSO"_4 * x"H"_2"O"#
we use
More often than not,
Now, the difference between hydrated copper(II) sulfate and anhydrous copper(II) sulfate, except for the fact that the former contains water of crystallization, is the color.
For example, copper(II) pentahydrate,
This means that when copper(II) sulfate pentahydrate is heated, its color will change from blue to white as the water of crystallization evaporates.
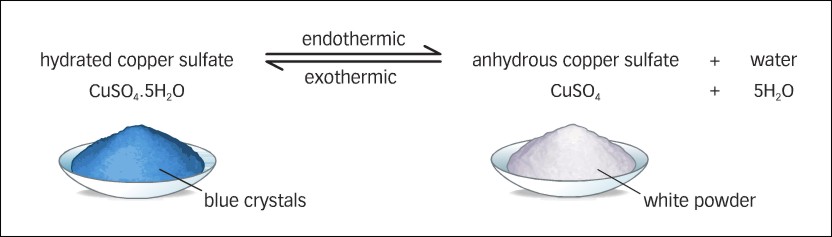
The reaction is reversible, i.e. if you add water to the white anhydrous copper(II) sulfate you'll get the blue copper(II) sulfate pentahydrate again.

The solid turns white.
Explanation:
The equation for the reaction is

(From www.yooniqimages.com)
In hydrates,
You probably started with the pentahydrate,
The hydrate is blue because the copper is present as the blue tetraaquocopper(II) ion,
As you heat the blue hydrate and drive off the water, the ion is converted to the colourless copper(II) ion,
The dehydration actually occurs in steps.


