How does bond polarity affect the strength of an acid?
1 Answer
Mar 18, 2014
Increasing the polarity of an
Explanation:
When shared electrons in an
It also becomes weaker.
It is easier for the proton to leave the molecule, so the compound becomes more acidic.
The electronegativity of elements in the same row of the Periodic Table increases from left to right.
Thus, the polarity and the acidity of the
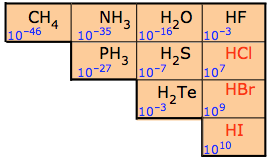
The chart below shows how acid strength increases from left to right across a row and from bottom to top in the Periodic Table