How does mass number relate to the particles in the atom?
1 Answer
Each element has a specific mass number and a specific atomic number.These two numbers are fixed for an element.
The mass number tells us the number (the sum of nucleons) of protons and neutrons in the nucleus of an atom.
The atomic number (also known as the proton number) is the number of protons found in the nucleus of an atom.
It is traditionally represented by the symbol Z.

Mass # of Carbon is 12, and its atomic number is 6.
A Carbon atom has atomic # 6 , so it has 6 protons in its nucleus.
it has mass number 12 , it means that the sum of neutrons and protons in a carbon atom is 12.
n + p =12
p =6
n +6 = 12
n+6-6 = 12-6
n=6.
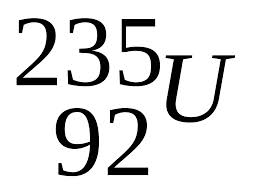
Mass # of Uranium is 235, and its atomic number is 92.
An Uranium atom has atomic # 92 , so it has 92 protons in its nucleus.
it has mass number 235 , it means that the sum of neutrons and protons in a carbon atom is 235.
n + p =235
p =92
n +92 = 235
n+ 92-92 = 235-92
n = 143

