How does the movement of particles in a stick of butter differ from the movement of particles in a dish of melted butter?
1 Answer
Dec 9, 2015
They move more in the melted butter.
Explanation:
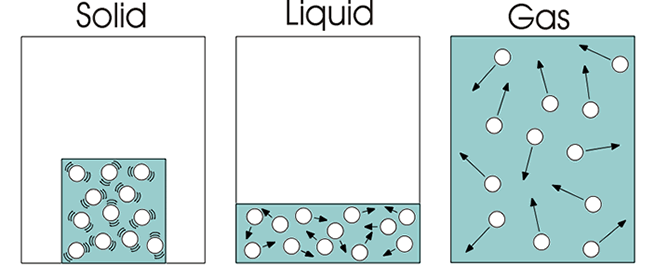
In every state of matter exists interactions between it's particles. This particles can move in many different ways: vibrating, rotating, translating, etc.
If you have a stick of butter you can see that it's, relatively, stiff and that's because the interactions between the butter particles are strong enough to hold the stick together and don't move.
When you apply energy to the butter stick and melt it, this energy applied is used to break those strong interactions so now the particles can move more.
 http://www.msm.cam.ac.uk/SeeK/slg.html
http://www.msm.cam.ac.uk/SeeK/slg.html

