How I can find the saponification of herb?? I need your valuable advises.
2 Answers
Are you asking about the preparation of herbal soaps?
Explanation:
Are you asking about the effect of the saponification process on the herbal contents?
Short answer: Most of the herbal compounds will survive the saponification process.
Saponification is the reaction of a triglyceride (fat) with a strong base to form glycerol and a mixture of fatty acid salts.
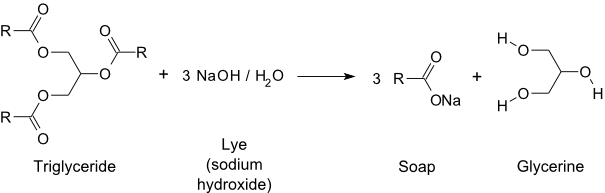
The reaction mixture is strongly basic.
Now let's look at herbs.
Herbs generally have a high flavonoid (antioxidant) content.
Most flavonoids are quite stable to alkali and heat, so they should be fine in the soap-making process.
Some herbs contain vitamins, and some vitamins are less stable to heat and the highly basic environment.
Herbs containing essential oils should probably be added late in the soap-making process, when the mixture starts to thicken.
Although the oils are probably stable, they are also volatile, and the heat of the reaction will cause them to evaporate and escape into the atmosphere rather than stay in the soap.
Here is a short presentation on herbal soap making.
Herbs do not contain enough fats to be directly "saponifiable".
Explanation:
See also: http://chemistry.about.com/od/cleanerchemistry/ss/How-Saponification-Makes-Soap.htm.
IF you mean how to make an herbal soap, you can mix finely ground herbs into the soap solution before it sets up to a solid to complete the process.


