How many molecules are in 31.9 g of hexane?
1 Answer
I will assume you mean
Multiply Avagadro's Number times the amount of hexane and divide by the molar mass of hexane.
(6.022 X
Explanation:
Note: If you were talking about Cyclohexane, the formula would be
The formula for obtaining the number of particles given the amount of sample in grams follows this formula:
(6.022 X
In performing a unit conversion, you are actually doing the same thing to the units that you are doing to the values.
(particles / mole) * (g) / (g/mole)
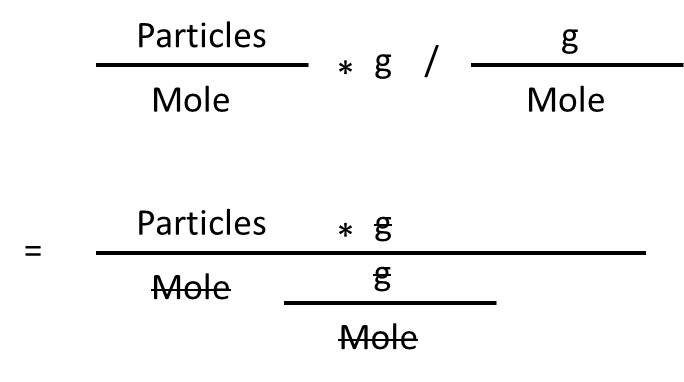 Source: Generated by David M. Using Microsoft PowerPoint
Source: Generated by David M. Using Microsoft PowerPoint
The moles and the grams cancel, leaving an answer in particles.
You may ask why I say "particles" and not "molecules"? The reason is so the SAME calculation may be performed for ionic compounds, ions, or other types of chemical identities.

