How many moles of argon are in 452 g of argon?
1 Answer
Dec 17, 2015
The number of moles in 452 grams of Argon is 13.31 moles.
Explanation:
In order to convert a substance from grams to moles, you must use that substance's molar mass. In this case, Argon's molar mass can be found using the periodic table. You can round however you like, but I am using 33.45 g/mol.
So start with what you are given: 452 grams of Argon. As mentioned before, you use the molar mass to convert from grams to moles. So take 452 grams of Argon and multiply by the molar mass of Argon. Your units will cancel out, leaving you with moles of Argon. 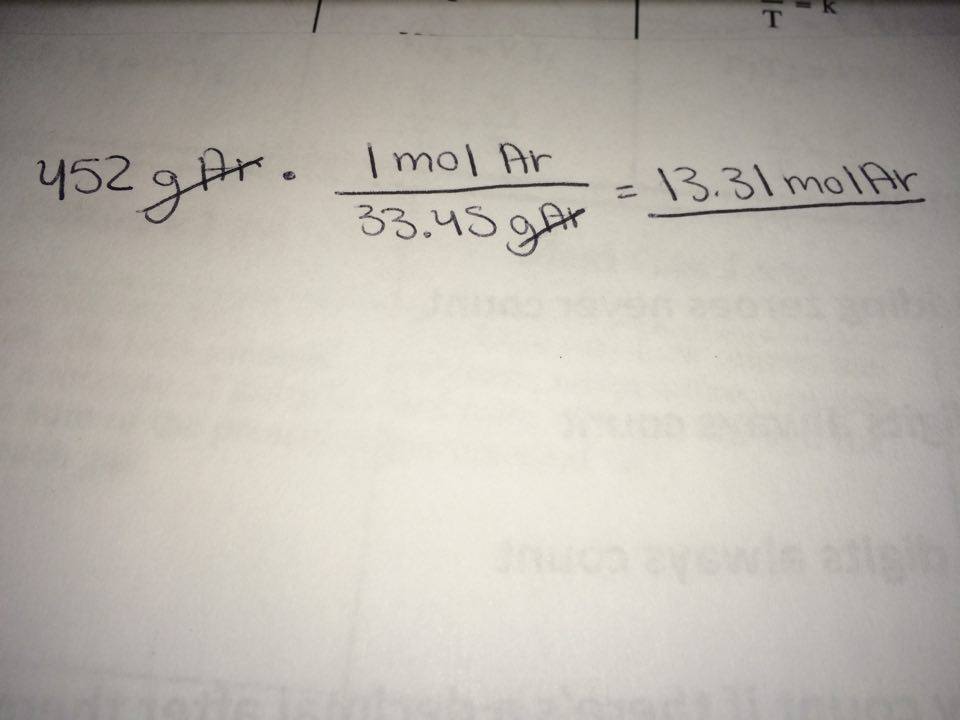
I hope that was helpful.

