How many more grams of KI will dissolve in 100 g of water at 40 °C than at 20°C?
1 Answer
Here;s what I got.
Explanation:
Your tool of choice here will be the solubility graph for potassium iodide,
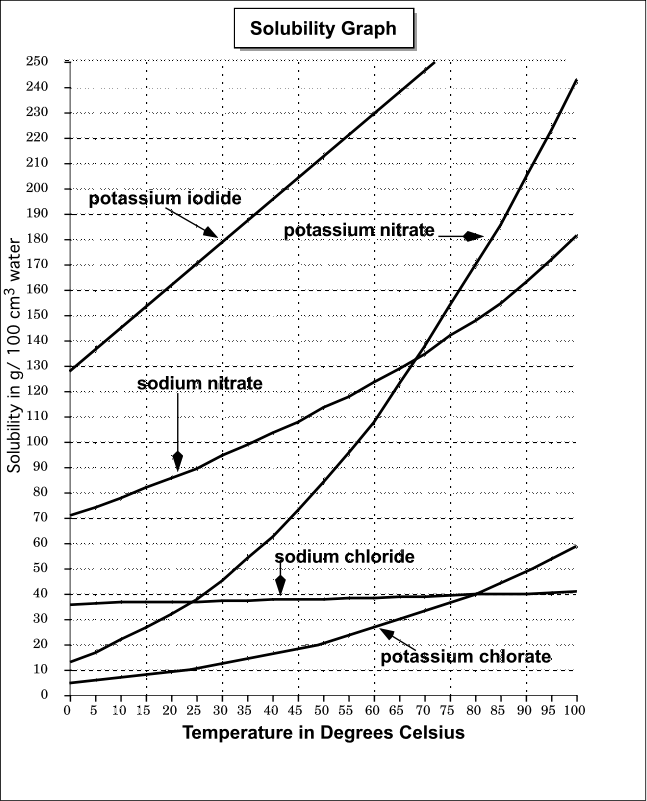
Now, this solubility graph expresses the solubility of potassium iodide per
Notice that at
At
These values can be found by tracing vertical lines starting from
Therefore, you can say that a saturated solution of potassium iodide will hold
That is the case because
#overbrace("195 g")^(color(blue)("solubility at 40"^@"C")) - overbrace("155 g")^(color(purple)("solubility at 20"^@"C")) = "40 g"#

