How do you find formulas for covalent compounds?
1 Answer
Normally, if the elements bonded are non-metal, that is a covalent bond.
Explanation:
Nonmetals are not big on the give-and-take department but they can share electrons with other nonmetals to satisfy the octet rule. Why? It has something to do with their placement in the periodic table (they are too close to each other, hence, the difference in their electronegativities are between 0 and 1.6).
Based on the periodic table, there are seventeen elements that are classified as nonmetals.
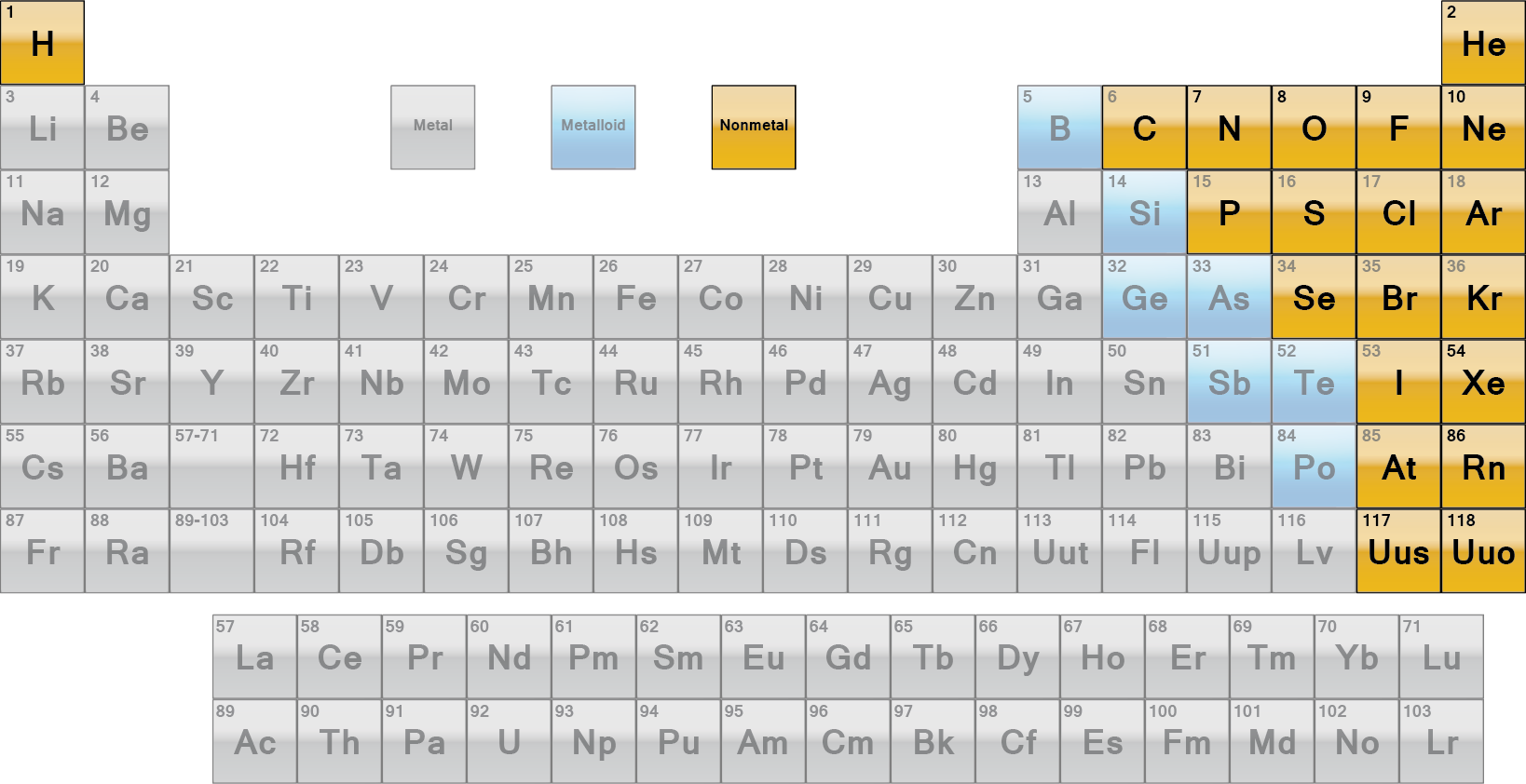
Hence, in a substance say, lead sulfate
1.) ionic bonding between the metal
2.) covalent bonding between the
Another thing that you can use is the electronegativity values to predict what kind of chemical bond would the elements undergo to.
If the difference in electronegativities between two bonding elements is less than 0.5, then you have a nonpolar covalent bond.
If the difference in electronegativities between two bonding elements is between 0.5 and 1.6, then you have a polar covalent bond.
If the difference in electronegativities between two bonding elements is greater than 2, then you have an ionic bond.
Examples: H= 2.1 ; Cl= 3.0; Na= 0.9; C= 2.5

