How will you convert 2 methylpent-2-enal to 2methylpentanal?
2 Answers
H2/Pt at 1 atm and room temperature (Corrected Answer)
Explanation:
You can turn an alkene into an alkane using hydrogenation. Basically what is happening is you are breaking the double bond and replacing that with two hydrogens, one on each side.
You can probably do that with H2/Pt at 1 atm and RT. Alkenes are much more easily reduced with H2/Pt than carbonyls.
NOTE: Pt (Platinum) is the catalyst of this reaction, and there are other catalysts one could use, but Pt is one of the most common ones.
Hope this helps (c:
How about this?
Explanation:
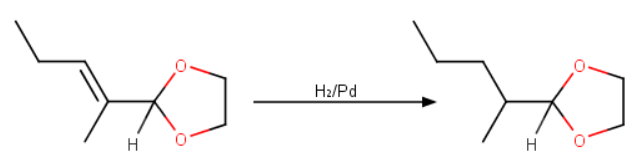
It is tempting to use hydrogen with a metal catalyst, but that will reduce both the alkene and the aldehyde group.
Instead, you must use a protecting group on the aldehyde to prevent it from being reduced.
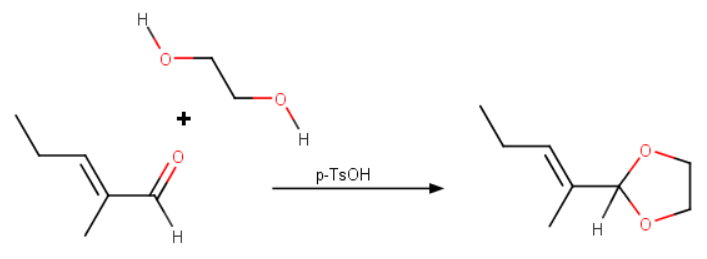
One way to protect an aldehyde is to convert it into an acetal by reaction with an alcohol and acid.
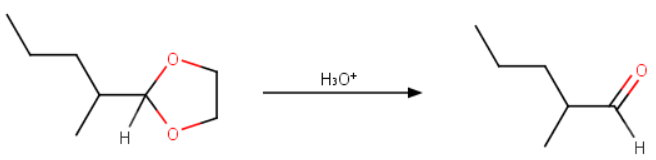
When you no longer need the protecting group, you can restore the aldehyde by adding aqueous acid.
Step 1. Adding the protecting group
React the aldehyde with ethylene glycol in the presence of p-toluenesulfonic acid.
Step 2. Reduction
Use hydrogen and a
Step 3. Remove the protecting group
Add aqueous acid.