How would you compare diffusion with effusion?
1 Answer
Here's my interpretation.
Explanation:
Diffusion is when a substance disperses evenly throughout a medium; i.e. a perfume dispersing its scent through a room:
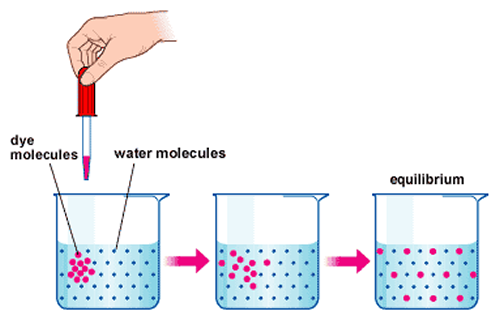
Effusion is when a substance "escapes" through a (tiny) opening; i.e. helium in a balloon escaping through the balloon's rubber network:
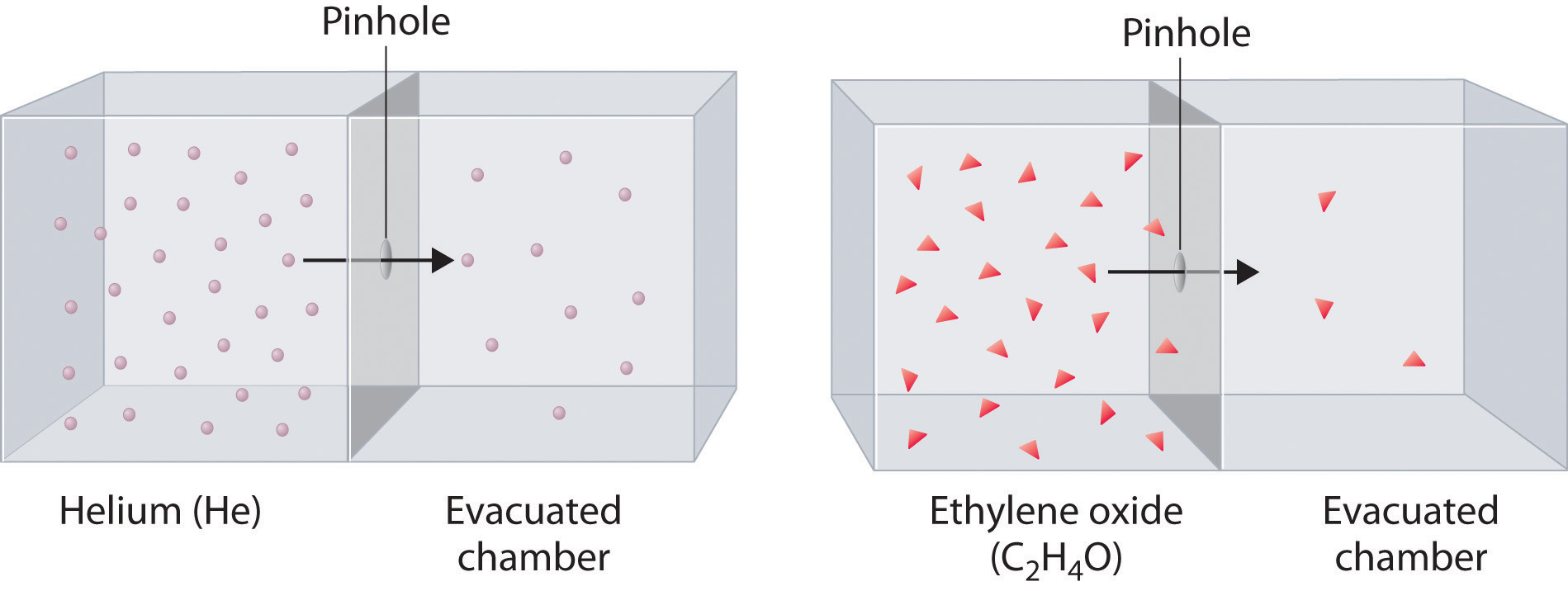
We can infer that the rates of diffusion and effusion of a gas depend on the speed of the particles.
In the effusion image, we notice that
Graham's law of effusion (or diffusion) explains this:
where
-
#(r_1)/(r_2)# is the ratio of the rates of effusion of a gas 1 and a gas 2 -
#M_1# and#M_2# are the molar masses of gases 1 and 2
This equation tells us that
The rate of effusion (and diffusion) of a gas is inversely proportional to the square root of its molar mass.
Therefore, the lower the mass of a gas, the higher its rate of effusion and the higher its speed.
This makes intuitive sense, as a gas with less mass should move faster than a gas with more mass.
In terms of rates and speeds, effusion and diffusion are quite similar.
The difference between the two is that diffusion is the spreading out of a substance within a dispersing medium, and effusion is when a substance escapes through a tiny pinhole, or other hole.
Another way to differentiate between diffusion and effusion is to imagine effusion as motion of particles through a hole one-dimensionally (i.e.
#+-x# ), and diffusion allows for all three dimensions (i.e.#+-x, +-y, +-z# ).
