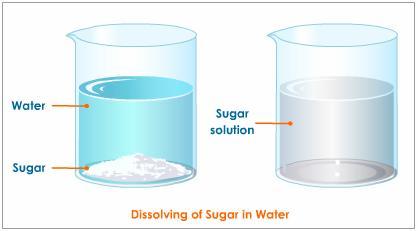
How would you identify the solute and solvent in this solution:. table sugar (#C_12H_22O_11#) in water?
1 Answer
Explanation:
The thing to remember about a solution is that you can distinguish between the solute and the solvent by simply looking at how much of each you have present in the solution.
Simply put, a solution will always contain more particles of solvent than particles of solute. The solvent will thus be present in larger amounts when compared with the solute.
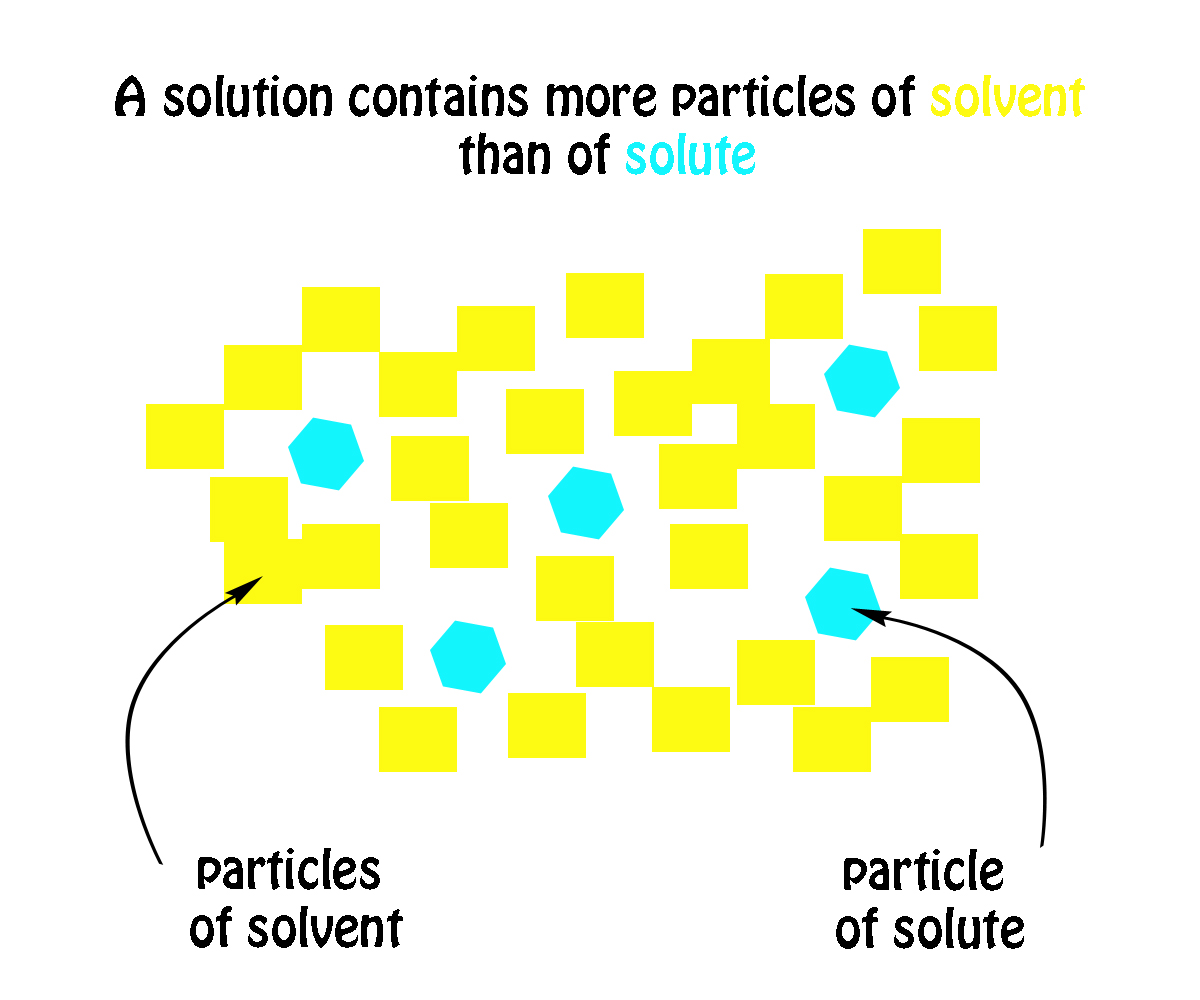
Another thing to remember here is that the solute is being dissolved in the solvent. In other words, the solute is being dissolved and the solvent does the dissolving.
In your case, sucrose,
Right from the start you should be able to say that because the sucrose is being dissolved in water, it will act as solute. Consequently, water will act as the solvent.