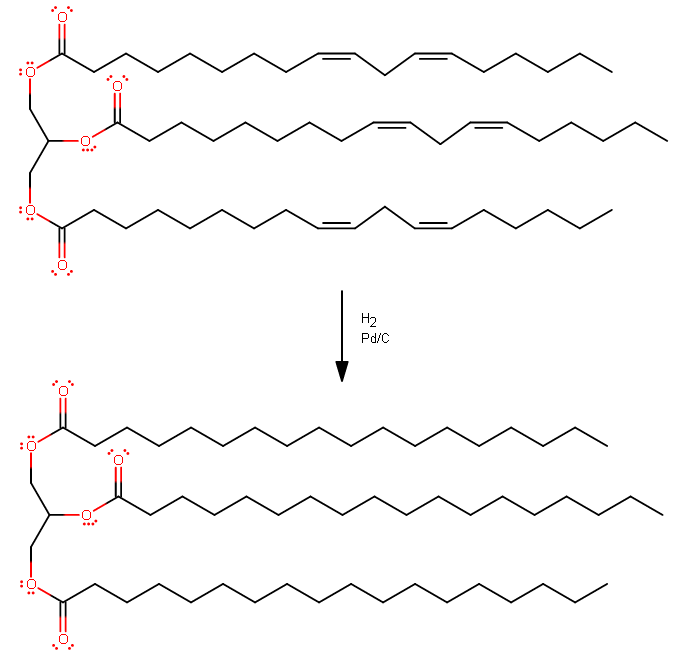
How would you write an equation for the hydrogenation of glyceryl trilinolenate?
1 Answer
Jan 12, 2016
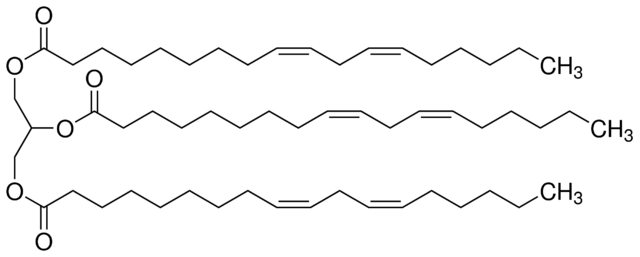
Linolenic acid is an 18-carbon carboxylic acid with three double bonds somewhere on it, so we know it has 18 carbons, including the carbonyl carbon.
This compound is a derivative of glycerol, so it has to have three carbons on the main chain (middle left):
I'm assuming full hydrogenation, because there is no reason why a particular double bond in this compound would be picked over another.
So, basically just take out all the double bonds. Adding