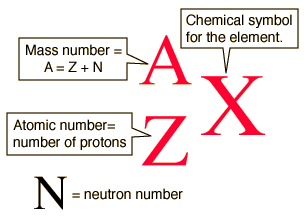How would you write the symbol for carbon -14 including atomic mass and number?
1 Answer
Dec 8, 2015
The nuclear notation of carbon-14 is
Explanation:
This is called isotopic notation or nuclear notation. You write the chemical symbol for the element, then write the mass number as a superscript on the left side, and the atomic number as a subscript on the left side.
Carbon-14
Mass number
Atomic number (from the periodic table)
Nuclear notation: