Identify the equation for which #K_c = ["Ni"^(3+)]["Cl"^-]^3#.?
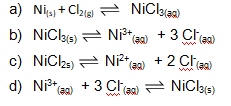
1 Answer
Choice b)
Explanation:
A reaction with forumla
will have an equilibrium constant of
Both
The number of moles of individual species shall equal to the respective powers of its concentration in the equilibrium constant expression. Thus in this question, there's one mole of
and three moles of
The concentration of pure solids and liquids does not affect the equilibrium position.[1] Therefore these species are assigned activity values of
References
[1] Clark, Jim. “Writing Equilibrium Constant Expressions Involving Solids and Liquids.” Chemistry LibreTexts, Libretexts, 21 July 2016, chem.libretexts.org/Core/Physical_and_Theoretical_Chemistry/Equilibria/Chemical_Equilibria/Calculating_An_Equilibrium_Concentrations/Writing_Equilibrium_Constant_Expressions_involving_solids_and_liquids .

