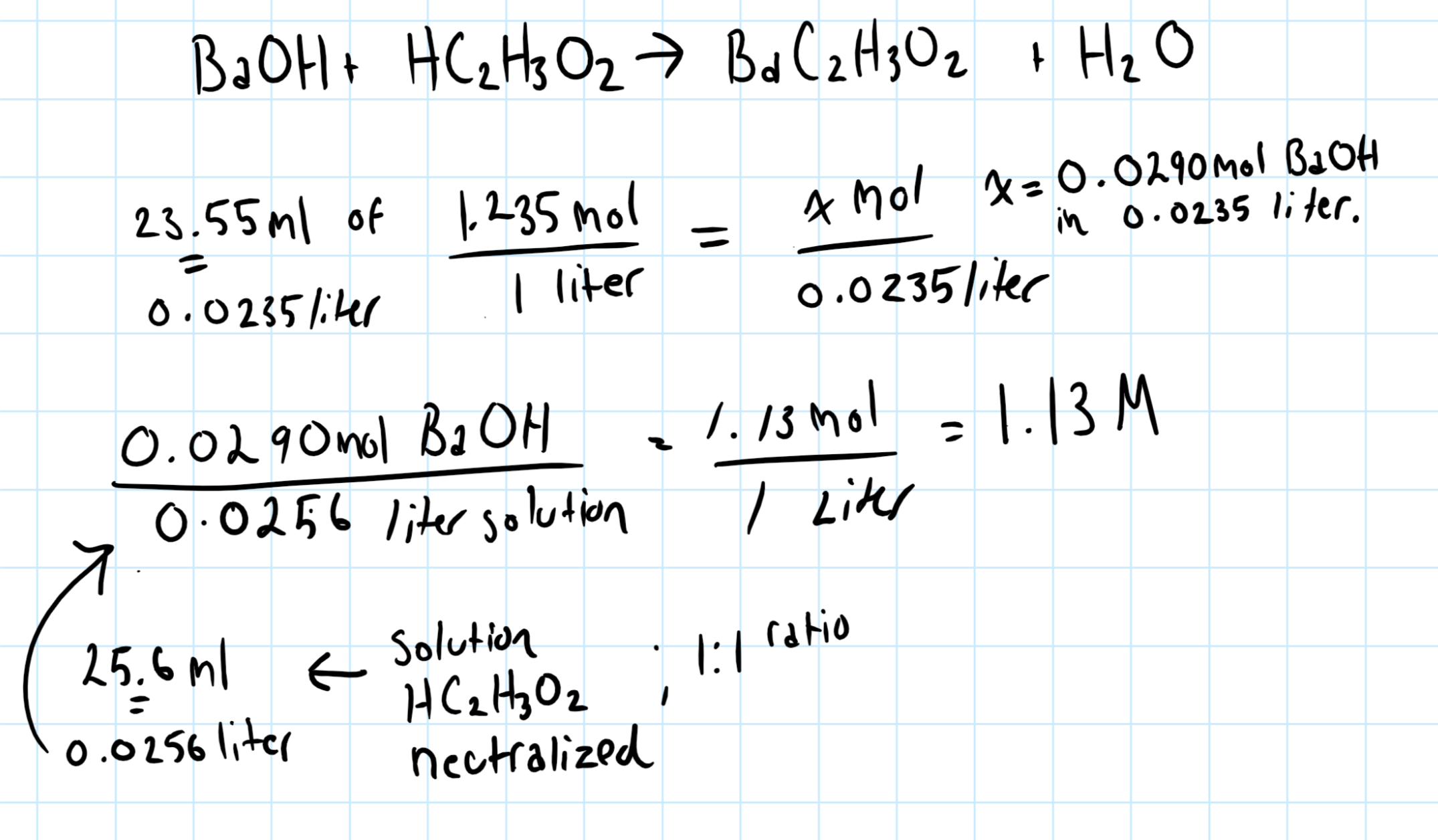
If 23.55 mL of 1.235 M barium hydroxide is required to fully neutralize a 25.6 mL solution of acetic acid, what is the concentration of the acid solution in M?
1 Answer
Dec 6, 2016
1.13M
Explanation:
In the graphic below the problem is done stoichiometrically.
However, for a quick and easy answer the
Consider: