Is octane a polar molecule and does it have an attraction for water?
1 Answer
No, octane is a non-polar molecule.
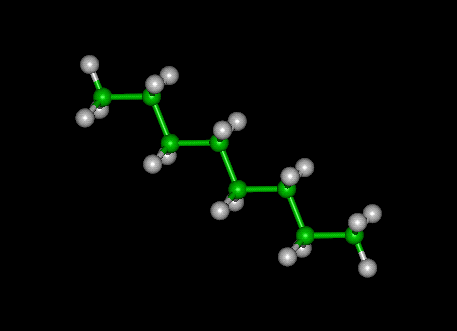
Octane (
Bond polarity is determined using the electronegativity values of the two atoms that form the bond. According to the Pauling scale,
In order for a bond to be considered polar, the difference in electronegativity between the two atoms must be greater than 0.5 (this will vary in some textbooks). Since this is not the case for
Since we've established that octane is considered to be non-polar, it will not be soluble in water, since water is a polar solvent. This will happen because octane (hydrocarbons in general) contains neither ionic groups, nor polar functional groups that can interact with water molecules. Such compounds are called hydrophobic (literally, water-fearing).
Here's an example of non-polar molecules in water, showcasing hydrophobic behaviour:
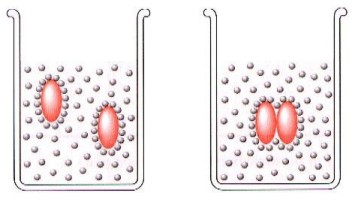
Non-polar molecules will stick together in order to minimize their contacts with the water molecules.

