Is temperature directly proportional to pressure?
1 Answer
Mar 20, 2018
Well, in a very special case.
Explanation:
Gas laws are extremely useful in experimental science, as they allow us to work out volumes, temperatures, pressures, and other variables for an ideal gases, even though no ideal gases exist.
Anyways, we can use an equation that connects temperatures and gases together, which is also known as Gay-Lussac's Law.
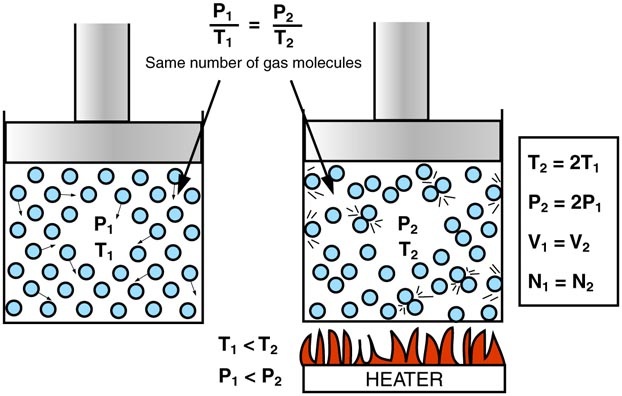
Gay-Lussac's Law: If the number of moles and the volume of a gas is constant, then temperature is directly proportional to pressure, i.e.
It might be useful to look at a diagram of the law: