My question is from a chemistry assignment Describe the structure and bonding associated with C3 of pent-2-ene? Thanks in advance!
1 Answer
May 30, 2018
Here's what I find.
Explanation:
The structure of pent-2-ene is
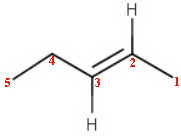
The important structural features are
-
Carbons 1, 4, and 5 are
#"sp"^3# hybridized, with a tetrahedral geometry and bond angles of 120° -
There is free rotation about the
#"C1-C2"# and#"C4-C5"# bond axes -
Carbons 2 and 3 are
#"sp"^2# hybridized with bond angles of 120° and a trigonal planar geometry -
Carbons 1, 2, 3, and 4 and the
#"H"# atoms on carbons 2 and 3 are all in the same plane -
There is no free rotation about the
#"C2-C3"# bond axis -
The
#"C=C"# double bond consists of a σ bond (#"sp"^2"-sp"^2# end-on overlap) and a π bond (#"p-p"# sideways overlap).

