The isotope Francium 224 has a half life of 20 minutes. A sample of the isotope has an initial activity of 9-- disintegrations s^-1. The approximate activity of the sample an hour later will be what? Multiple choice question. Please explain why for answer
1 Answer
May 12, 2017
The number of disintegrations is
Explanation:
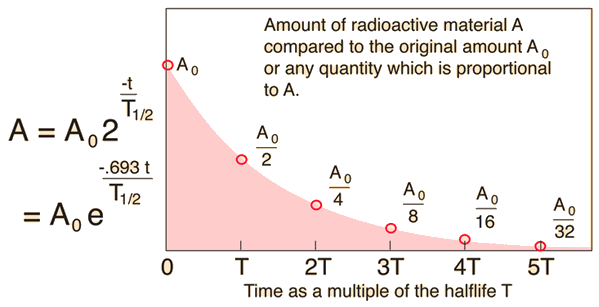
The half life is
The time is
So, we have
The number of disintegrations is

