The mass of a proton is #1.67*10^(-27)# kg. What is the mass of a proton in picograms?
1 Answer
Mar 29, 2015
The mass of a proton in picograms is
This is a basic unit conversion problem in which you must go from one order of magnitude, in your case kilograms, to another order of magnitude, picograms.
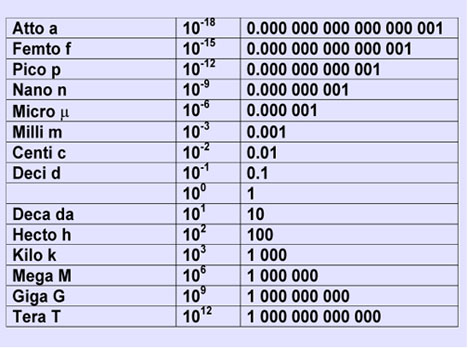
As you can see, 1 kg contains
So, set up two conversion factors to take you from kg to grams, and then from grams to picograms

