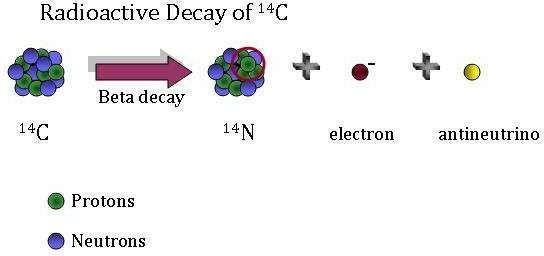
To determine the age of fossils it is possible to analyze the amounts of the isotopes #"^14C# and #"^14N# because over time the #"^14C# - which originated in the atmosphere - breaks down into #"^14N#. What net change occurred for this to happen?
2 Answers
One neutron in
Explanation:
Carbon 14 is not a stable isotope. It has more neutrons that are needed to stabilize the repulsion of the six protons in the Carbon Nucleus. One of the extra neutrons breaks down into a proton and electron.
Looking closely at the equation there is no net change. The positive charge on the proton balances the negative charge on the electron.
net charge remains zero
Again looking at the equation there is no net change in the mass. The neutron has a mass of one and the sum of the mass of the proton and the electron is also a mass of one.
Note because of the relatively short half life of Carbon 14 only very recent fossils can be dated using Carbon 14. Any thing older than 50,000 years can not be accurately dated using Carbon 14. A textbook I taught out falsely claimed that dinosaurs supposed to be more than 65 million years old had been dated using Carbon 14.
The net change was that a proton changed into a neutron and an electron.
Explanation:
The overall reaction was that the carbon-14 experienced β-decay.
The net reaction was that a neutron in the nucleus of a carbon-14 atom emitted an electron and changed to a proton and an electron antineutrino.