Two charges of # 2 C # and # 3 C# are positioned on a line at points # -5 # and # 6 #, respectively. What is the net force on a charge of # -8 C# at # -2 #?
1 Answer
Let us use Coulomb's Law for electrostatic interaction.
Explanation:
Charges distribution is given by this picture:
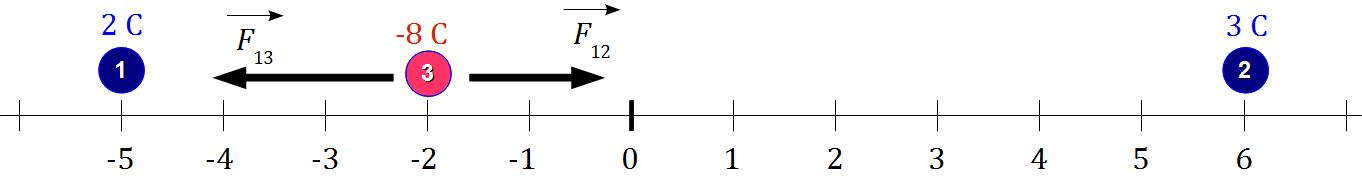
where blue charges will be our fonts, and the red charge will be our object charge.
According to Coulomb's Law, the force between two charges 1 and 2 is given by:
where
Electrostatic force is repulsive if both charges have the same sign, and attractive if they have opposite signs.
Let us calculate both forces:
- Force from 1 to 3:
- Force from 2 to 3:
Both forces are attractive (that's why both signs are negative). We must substract them, and the resultant force has the same orientation than the bigger one:

