The formation reaction is an hypothetical reaction in which any substance is formed from its component elemental substances in their most stable thermodynamic state.
Being urea the crystal molecular compound CH₄N₂O,
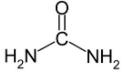
the formation reaction equation is:
#C(graph) + 2H_2(g) + N_2(g) + 1/2O_2(g) -> CH_4N_2O(s)#
The energy change #Δ_fU°# associated to the formation of one mole of urea cannot be calculated.
It is an experimental value.
A good source of this kind of data is the NIST Webbook, at this link
There you can get the most updated experimental data of -333.11 kJ/mol for the standard enthalpy of formation #Δ_fH°#.
This variation of enthalpy is not much different from the wanted standard variation of energy #Δ_fU°# because the enthalpy change is defined as the energy change due only to the absorbed heat in the chemical system (that is #ΔU# subtracted from the compression work #-PΔV# entering the system at constant pressure).
So, from the "definition" of #ΔU# given by the first principle of thermodynamics, #ΔU = Q + W#, we get #ΔU - W = Q = ΔH#. By substituting #W = -PΔV# we get: #ΔU = ΔH-PΔV#
The #PΔ_fV°# accompanying the formation of one mole of urea can be easily calculated from the ideal gas general equation:
#P*V=n*R*T -> PΔ_fV° =Δn*R*T = -3.5*8.314*298.15 = -8675.9 " J/mol" = -8.68 " kJ/mol"#
Where #Δn = -3.5" "mol# is given by the disappearing of two moles of dihydrogen gas, one mole of dinitrogen gas and half a mole of dioxygen gas to yield a negligible volume of solid urea (partially compensated by the disappearing of one mole of carbon graphite).
So we can conclude that
#Δ_fU° = Δ_fH°-PΔ_fV° =#
#=-333.11 "kJ/mol" -(-8.68 "kJ/mol") = -324.43 "kJ/mol"#.
As usual, the difference between #Δ_fU°# and #Δ_fH°# is not so big, even in reactions with gases. And it is negligible in reactions with reactants and products in the condensed phases only.


