What are the cleansing effects of soaps?
1 Answer
Jan 22, 2017
Soaps are the salts of long chain fatty acids..........
Explanation:
A typical soap can be made from lauric acid,
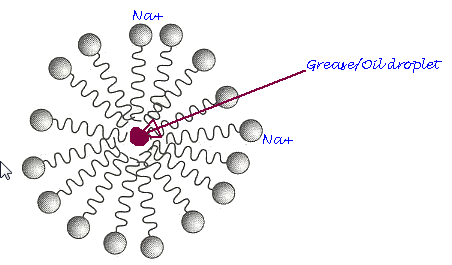
The soap, sodium laureate, has some water solubility due to carboxylate function; the grime and grease that the soap dislodges tends to be trapped in the hydrophobic tail of the soap, which agglomerates to form micelles.
Anyway, this will be explained better in your text, so please consult it.