What are the differences between alpha and beta particles?
1 Answer
Alpha particles are helium nuclei and beta particles are fast moving electrons.
Explanation:
Alpha Particles
Alpha particles are released when a nucleus is too heavy - this means there are so many protons and neutrons in the nucleus that the nucleus becomes unstable. The alpha particle consists of two protons and two neutrons, so it is essentially a helium nucleus, and this decreases the isotopes atomic mass by four and its atomic number by two, meaning it becomes a different type of atom.
For example, Radon-219 (atomic number 86) emits an alpha particle to become Polonium-215 (atomic number 84).
Note that 219 - 215 = 4 (from the mass of the 2 protons and 2 neutrons) and 86 - 84 = 2 (from the 2 lost protons)
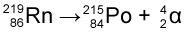 BBC Bitesize
BBC Bitesize
Alpha particles are the largest form of ionising radiation (radiation that knocks electrons off atoms when it collides with them) and are therefore the most ionising (how well the radiation collides with and knocks electrons off of atoms) - the bigger a particle is, the more likely it is to collide with atoms.
However, it is the least penetrating (again, because they are large particles they hit atoms quickly and get absorbed) and can be absorbed by just a sheet of paper. In air, it has a range of about 5cm.
Beta Particles
Beta particles are essentially fast moving electrons. They are produced when an atom has too many neutrons (there's some complicated quantum mechanics to explain why too many neutrons make an atom unstable - let me know if you are interested as it took me a very long time to work out!). To make the atom stable again, one of the neutrons changes into a proton. Because a proton is positive whilst a neutron is neutral, this means that somehow we need to find a way to account for the lost negative charge - so as well as a proton, the neutron also produces a high energy electron (think of the negativity of the electron balancing out the positivity of the proton to make a neutron neutral). This causes the atomic mass to remain unchanged (electrons have such a small mass that there is no point counting it for this - and yes, the fact they have some mass means that a neutron is slightly heavier than a proton) but the atomic number increases by one, because the atom has essentially gained a proton and lost a neutron.
An example is Carbon-14 (atomic number 6) releasing a beta particle to become Nitrogen-14 (atomic number 7).
Note that the atomic number (number of protons) has increased by one, and the mass has stayed the same.
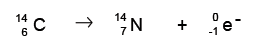 BBC Bitesize
BBC Bitesize
Beta particles are between alpha particles and the third type of ionising radiation - gamma rays - in terms of how strongly ionising they are. They are also in the middle for how penetrating they are and will pass through paper but be stopped by aluminium foil (a few millimetres thick). It has a range in air of around 15cm.
I hope this has helped! Let me know if I can do anything else, as this is one of my favourite areas of physics and so I would love to talk about it:)
