What are transition elements?
2 Answers
Jul 29, 2016
A transition element is an element whose d-block is never fully filled in its atomic state as well as in its ionic state .
Explanation:
- Elements of Group 3-11 are all transition elements.
The elements in the center of the periodic table
Explanation:
They are the elements towards the middle of the periodic table.
They are separated from the rest of the elements because their valence electrons are in both the
Transition elements are more strictly defined as elements whose atoms have an incomplete
This definition excludes elements in Group 12, as they have full
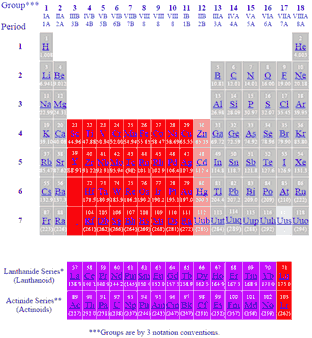
Thus,



