What determines whether a reaction is classified as sn1 or sn2?
1 Answer
See Explanation
Explanation:
It is good to know why they are called SN 1 and SN 2; in SN 2 reactions, the rate of the reaction is dependent on two entities (how much nucleophile AND the electrophile is around), and hence it is called SN2.
SN 1 reactions' rates are only dependent on on entity, the electrophile (loss of a leaving group is the first step of this reaction and it does not require a nucleophile at first to have the Leaving Group leave and form carbocation), therefore it is called SN1.
Now answering your question, here are how they differ and how you can determine which mechanism is happening:
1) The substrate (aka the electrophile): For SN2 reactions, a primary substrate is better, while a tertiary substrate is virtually unreactive. But for SN1 reactions, it is the opposite. Tertiary substrates are perfect for SN1 reactions and primary substrates are just not good!
Therefore, if you have primary or secondary substrates, then the reaction will proceed through SN2 mechanism. If you have Tertiary substrate, then it will proceed via SN1 mechanism.
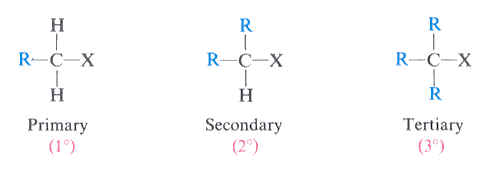
Where X is a leaving group (generally a halogen).
2) The Nucleophile: Since the rate of SN2 reactions are dependent on the electrophile AND the nucleophile, you need a strong nucleophile to speed up the reaction. SN1 reactions don't need a strong nucleophile.
Therefore, a strong nucleophile favors SN2, while a weak nucleophile allows for SN1 .
3) Lastly, the Solvent : Polar Aprotic solvents speed up rate of SN2 reactions. Examples include: Acetone, DMSO, DME, DMF. You see these in a reaction, it's SN2.
Hope this helps (c:
