What difference is there between magnesium chloride reacting with zinc nitrate or potassium sulfate?
My book says that if we take MgCl2 + K2SO4, there will be no reaction.
However, it does contain an equation for MgCl2 + Zn(NO3)2:
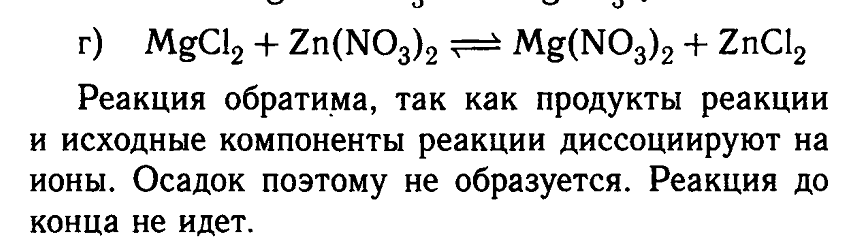
It mentions that the reaction is reversible, since both the products and the original components dissociate in water. "The reaction won't go to the end".
But is it different in any way from the reaction with K2SO4? It seems to me that we would have the same picture with it. However, the book flatly states 'no reaction'.
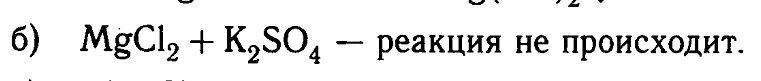
What is the difference?
My book says that if we take MgCl2 + K2SO4, there will be no reaction.
However, it does contain an equation for MgCl2 + Zn(NO3)2:
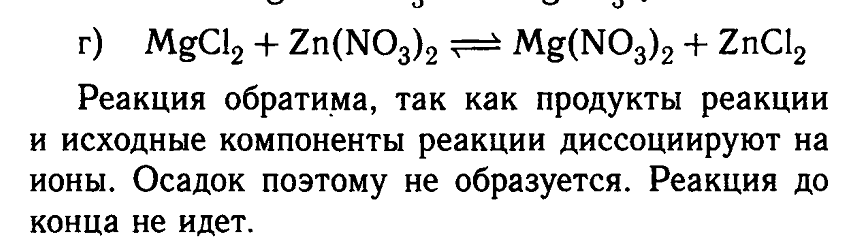
It mentions that the reaction is reversible, since both the products and the original components dissociate in water. "The reaction won't go to the end".
But is it different in any way from the reaction with K2SO4? It seems to me that we would have the same picture with it. However, the book flatly states 'no reaction'.
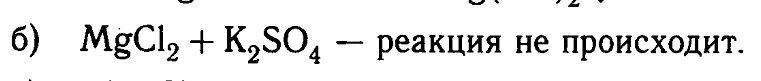
What is the difference?
1 Answer
In both cases there is no reaction.
Explanation:
For magnesium chloride and potassium sulfate all the ions are freely soluble so there is no reaction:
All the ions remain in solution.
The same situation occurs with magnesium chloride and zinc nitrate:
i.e no reaction as well.
I don't know why your text has written
Your text gives no state symbols so I have assumed

