What do you mean by a precipitation reaction?
1 Answer
See below.
Explanation:
A precipitate reaction means that during the reaction, solids form from an aqueous solution. Also, precipitation reactions occur when cations and anions in aqueous solution combine.
Here are some examples,

Here you can see that lead(II) cations and iodide anions are the products of the aqueous solution. Looking at the solubility chart, you can see that lead(II) cations and iodide anions do in fact make a solid and also that the sodium cations and nitrate anions are aqueous (as sodium nitrate is soluble).
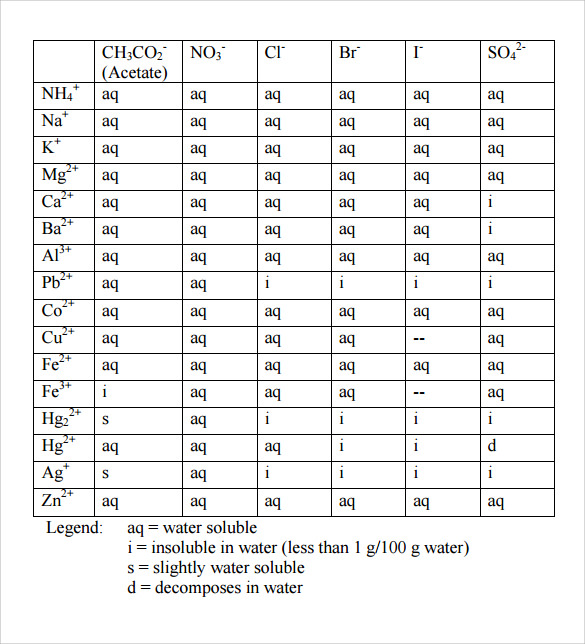
The Solubility Chart

An example of a precipitate


