What does crude oil consist of? What does fractional distillation do to crude oil?
1 Answer
Crude oil is raw material extracted from the ground, which consists of many different hydrocarbons. Fractional distillation is the process of separating these different hydrocarbons.
Explanation:
Crude Oil:
-
Crude oil is a mixture of hydrocarbons that exists as a liquid in natural underground reservoirs and remains a liquid when brought to the surface.
-
You will often hear oil referred to as petroleum. However, petroleum is a collective term that includes crude oil, condensate and gas.
-
The various forms of crude oil are extracted from the earth using a drilling process, and then further refined into fuels such as petrol, kerosene, diesel and many heavy waxes.
-
Petroleum plants can use the process known as cracking to reduce the size of very large hydrocarbon molecules in to more applicable molecules for every day life.
Fractional Distillation:
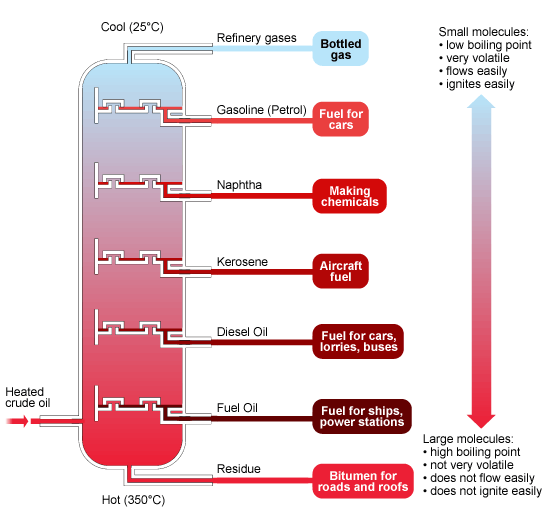
- As the different components in crude oil have different boiling points, the substances in crude oil can be separated using fractional distillation.
- The crude oil is evaporated and its vapours allowed to condense at different temperatures in the fractionating column. Each fraction contains hydrocarbon molecules with a similar number of carbon atoms.
-
Hydrocarbon molecules with a higher molecular weight have stronger dispersion forces which subsequently increases their boiling point. For example fuel oil.
-
Hydrocarbon molecules with a lower molecular weight have weaker dispersion forces which subsequently decreases their boiling point. For example refinery gases.