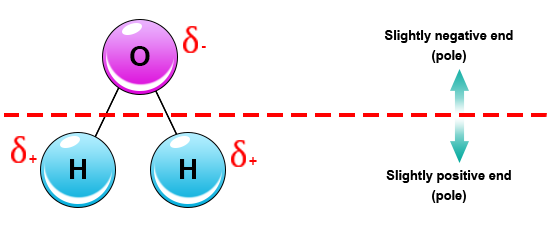
What does it mean when we say that water is a polar molecule?
1 Answer
Oct 17, 2016
This means that in the water molecule, one side is positively charged and the other side is negatively charged.
Explanation:
Since the water molecule (comprising two atoms of Hydrogen and one of Oxygen) is formed by covalent bonds, the electrons are shared.
This sharing causes the electrons to stay closer to the oxygen atom giving it a slight negative charge while the hydrogen atoms, as a consequence, have a slight positive charge.