What happens during nuclear transmutation?
1 Answer
May 4, 2016
In the transmutation processes, an atom has its nucleon attributes changed (e.g. number of protons)
Explanation:
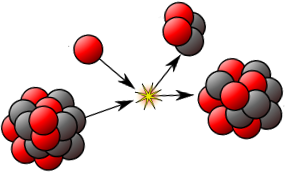
Nuclear transmutation is the conversion of one chemical element or an isotope into another.
A transmutation can be achieved either by nuclear reactions (in which an outside particle reacts with a nucleus) or by radioactive decay (where no outside particle is needed).
Therefore, in nuclear transmutation, an atom has its "identity" (e.g. atomic mass) "lost" (changed) due to nucleon alteration.
See (external links)
See (related Q&A)

