What happens to atoms during a chemical reaction?
1 Answer
Dec 2, 2015
They are rearranged to form new products with different physical and chemical properties from the reactants.
Explanation:
During a chemical reaction, the bonds between the atoms in the reactants are broken and the atoms bond in new ways to form products that are physically and chemically different from the reactants.
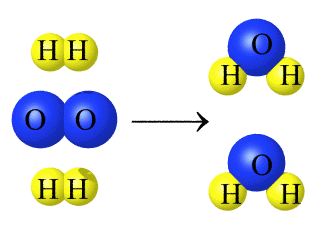
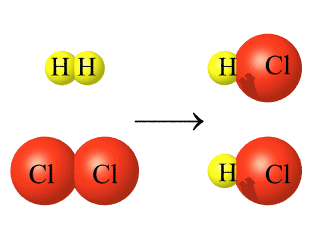
Regardless of the chemical change that takes place, the atoms in the products are the same atoms that were in the reactants, as demonstrated using color in the reaction below.
Formation of Hydrogen Chloride from Hydrogen Gas and Chlorine Gas
Formation of Water from Hydrogen Gas and Oxygen Gas