What happens to red blood cells placed into salt water?
1 Answer
Oct 8, 2016
The cells shrivel.
Explanation:
Salt water is a hypertonic solution in comparison to the internal cellular liquid, since there are more solute particles outside in the salt water than inside in the cytoplasm. This means that water will move out of the cells by osmosis due to the concentration gradient, and the cells will become shrivelled.
Here is a diagram demonstrating how animal cells react to different osmolarities.
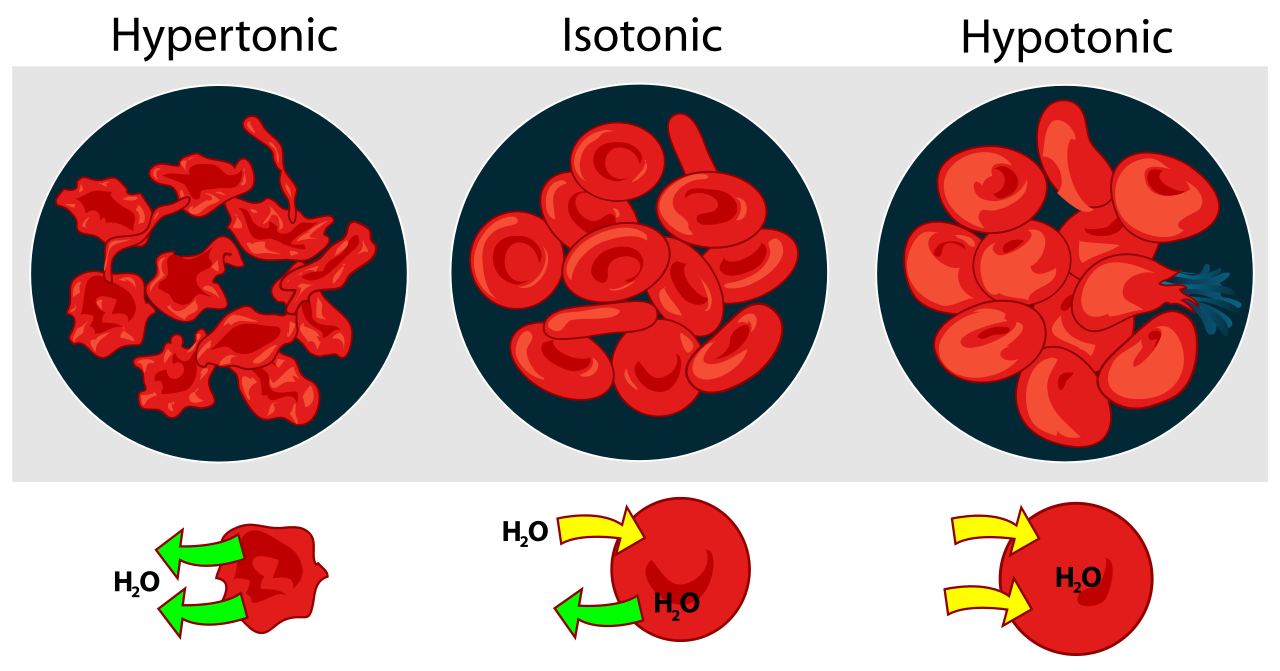
- Note that plant cells have cell walls, so instead of bursting in hypotonic solutions, they become turgid and the cell membrane pushes against the cell wall. Water no longer enters at this point becomes turgor pressure = osmotic pressure.

