What is a combination of two or more elements chemically combined?
1 Answer
May 13, 2017
A combination of two or more elements chemically combined is a compound .
Explanation:
Compounds can be molecular or ionic. In a molecular compound, the electrons of the elements making it up are shared, forming covalent bonds. The smallest particle of a molecular compound that has the properties of that compound is a molecule.
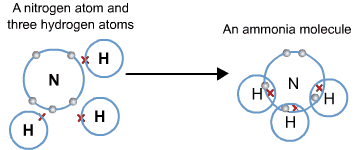
In an ionic compound, atoms lose or gain electrons and form positive or negative ions. Oppositely charged ions are attracted to one another and form an electrostatic attraction called an ionic bond. Ionic compounds make up a crystal lattice, so there is no discrete molecule. Instead, the formula consists of the smallest whole number ratio of ions, called a formula unit.


