What is a common characteristic of a metal?
1 Answer
Oct 10, 2016
Lustrous, malleable, ductile, electrically conductive
Explanation:
The metallic lattice consists of layers of metal cations being held together by a "sea" of their delocalised electrons. Here's a diagram to illustrate this:
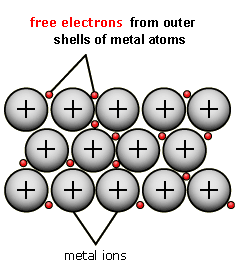 http://www.bbc.co.uk/schools/gcsebitesize/science/add_aqa_pre_2011/atomic/differentsubrev5.shtml
http://www.bbc.co.uk/schools/gcsebitesize/science/add_aqa_pre_2011/atomic/differentsubrev5.shtml
Lustrous- The delocalised electrons vibrate when hit by light, producing their own light.
Malleable/ductile- Metal sheets/ions are able to roll past each other into new positions, without the metallic bonds breaking apart.
Electrically conductive- delocalised electrons are able to move around and carry a current.

