What is activation energy?
2 Answers
Apr 28, 2017
It is the amount of energy needed to start a chemical reaction.
Explanation:
Some chemical reactions that are essential to life have activation energies that are too high or too slow to be of any use to the cell. Hence, catalysts are needed to lower activation energy so that a reaction can occur quicker. 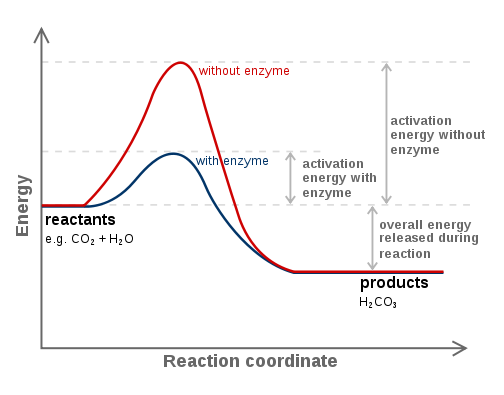
May 21, 2017
The energy needed to initiate a reaction.
Explanation:
The amount of energy which colliding particles must have in order to start a chemical reaction and change reactant particles into products.


