What is an example of an Arrhenius acid practice problem?
1 Answer
Jun 17, 2015
An acid is an Arrhenius acid so long as it dissociates into something and a proton.
Try this:
Identify which of the following 4 molecules would act as an Arrhenius acid:
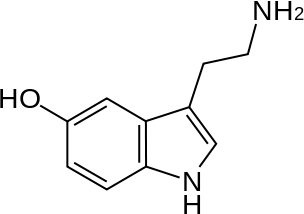
(Serotonin)
Hint: There are two, and an "acid" can have a pH of 7.

