What is an experiment in that all factors are identical except the one being tested?
1 Answer
It is a controlled experiment.
Explanation:
In a controlled experiment all variables are kept the same, except for the one being tested, which is called the experimental variable , or independent variable . This group is the experimental group . The group that is not affected by the experimental variable is the control group .
Very often a graph will be used to analyze the results of the experiment. The independent/experimental variable is placed on the x-axis and the dependent variable is placed on the y-axis. The dependent variable is what is measured as a result of the application of the experimental variable.
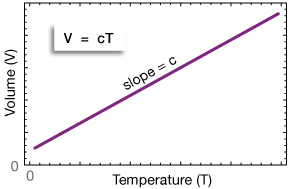
The graph above represents Charles' law. Charles' law states that the volume of a gas is directly proportional to the Kelvin temperature, with pressure and amount of gas to be held constant. Temperature is the experimental/independent variable because it can be varied, and volume is the dependent variable. The experimenter varies the temperature and measures the resulting volume. The slope of the line is the constant

