What is double bonding?
1 Answer
Double bonding occurs when two atoms of non-metal elements share two pairs of electrons, in what is called a double bond. A double bond is composed of two covalent bonds. In a structural formula, a double bond is represented by two parallel lines, each line representing a covalent bond between atoms.
Carbon atoms form double bonds ( C=C ) with each other in many organic compounds, which is why it is so versatile in the formation of organic compounds and biomolecules. The oxygen atoms in an
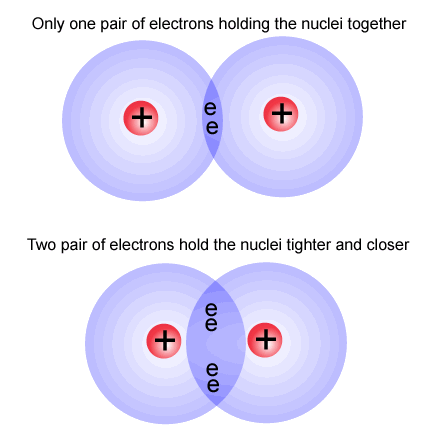
As you can see in this diagram the electrons are being shared between two atoms. The top part is a single bond and the bottom part is a double bond.
Image taken from: http://picornot.com/domain/ibchem.com


