What is the bond line notation for pentane?
1 Answer
Apr 19, 2018
See explanation
Explanation:
Bond line drawings are a simple way of drawing/writing the organic molecular structure.
Here is Pentane:
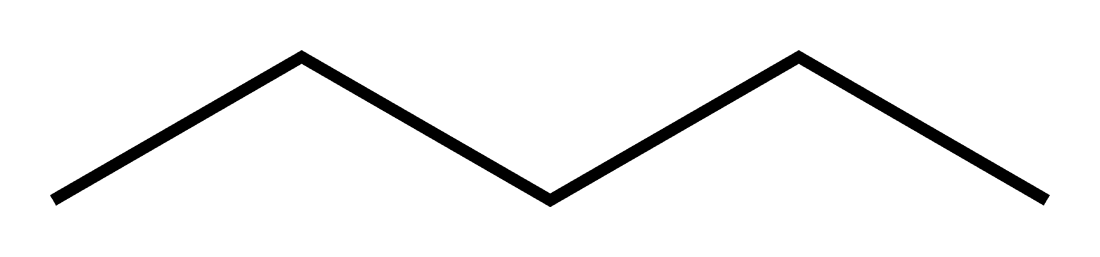
At the end of each line is a carbon. And each carbon has 2-3 bonds with hydrogens depending on where each carbon is.
The two carbons at the two ends of the structure are bonded to 3 hydrogens each, whereas the middle three carbons are bonded to 2 carbons each.
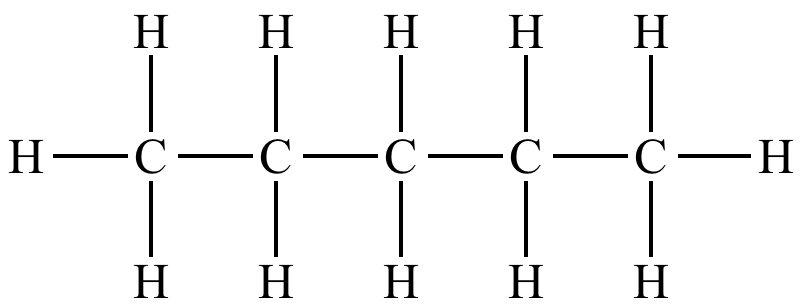
This makes it easier to imagine. 3 hydrogens at the end carbons and 2 hydrogens attached to middle carbons.
In bond line drawing of hydrocarbons, you only draw the carbons and not the hydrogens.
For example here is heptane:
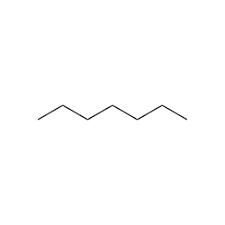
Hopefully you can see the pattern.
Hope this helps (c:
