What is the electricity required to produce 1 photon,red photon and blue photon?
1 Answer
I hope is not too confusing...
Explanation:
As an example consider the spectrum:
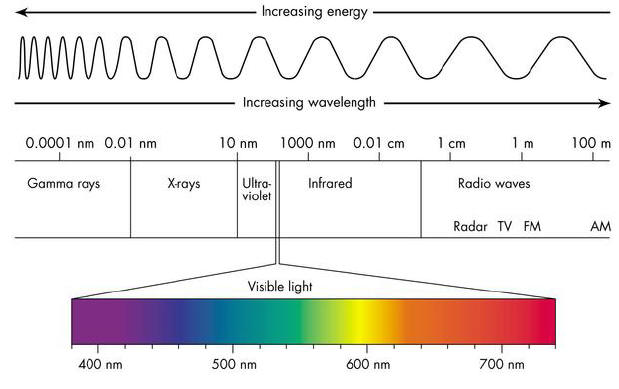
We can change the wavelength
so:
Blue light (roughly)
so we can find the energy needed to get one blue photon as:
Now if you have a light generator (hypothetical) you can feed one coulomb carrying this energy and it will produce one blue photon. In terms of current you can produce 1 blue photon each second if you send one of these coulombs of charge each second (representing a current of 1 Ampere).
So summarizing:
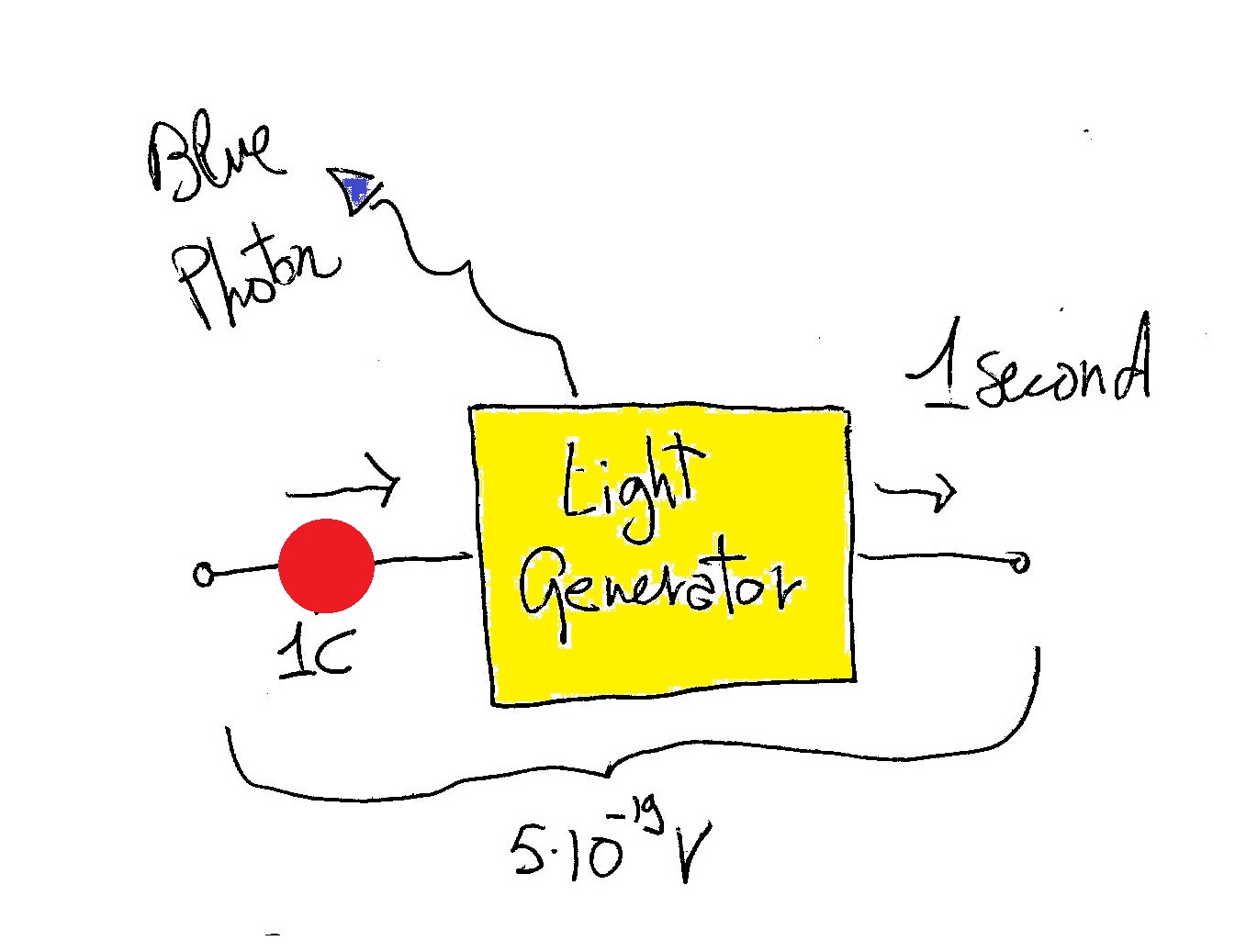
The real process of producing a blue photon could be simplified as:
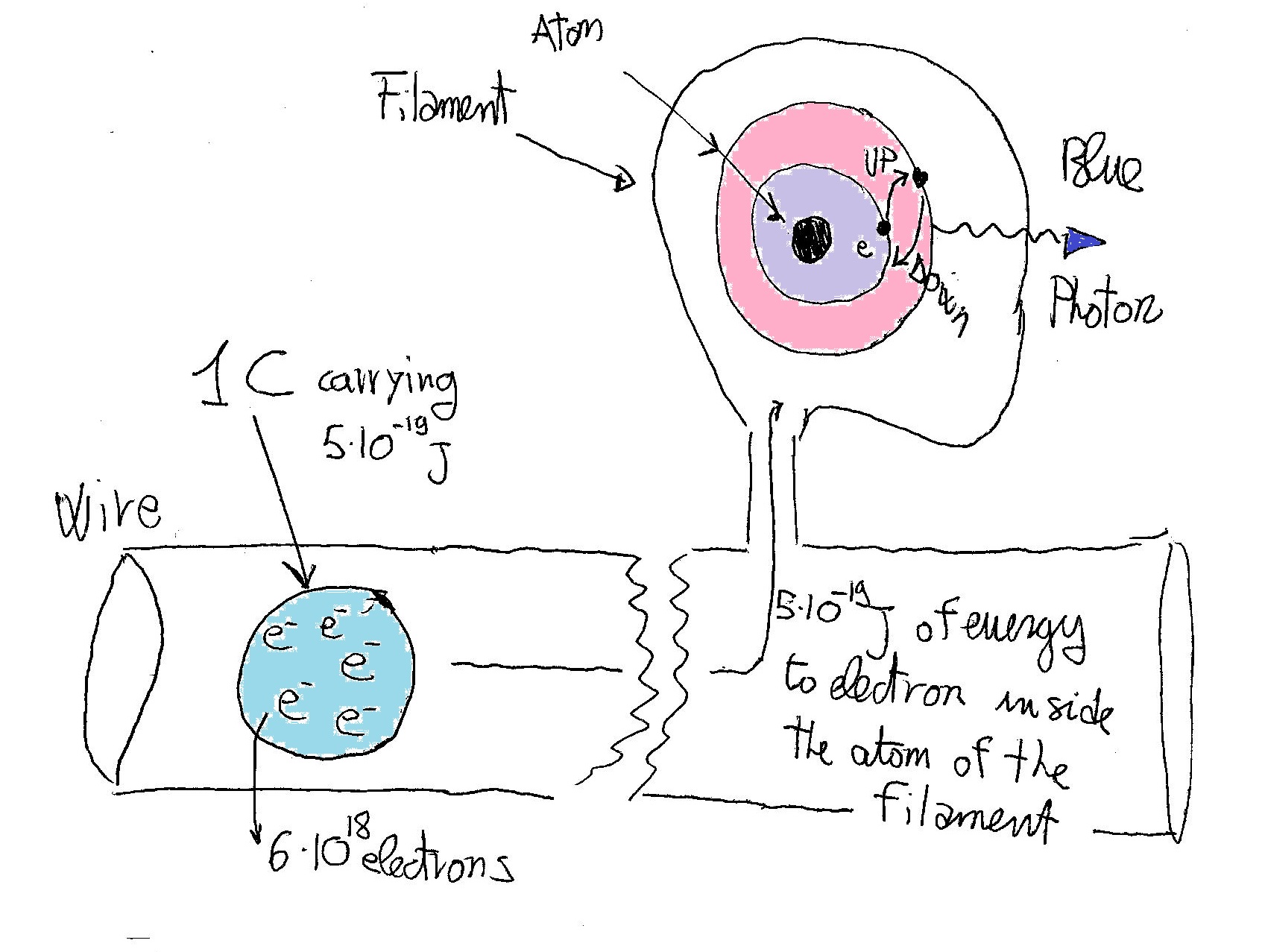
Inside the atom of the filament, the electron that "acquired" energy from the current and jumped to a higher orbital relaxes back emitting the surplus energy in form of a photon of light.

