What is the equation for finding the equilibrium constant for a chemical reaction?
1 Answer
We need to know two things in order to calculate the numeric value of the equilibrium constant:
The balanced equation for the reaction system, including the physical states of each species. From this the equilibrium expression for calculating Kc or Kp is derived.
the equilibrium concentrations or pressures of each species that occurs in the equilibrium expression, or enough information to determine them. These values are substitued into the equilibrium expression and the value of the equilibrium constant is then calculated.
Calculate the value of the equilibrium constant, Kc , for the system shown, if 0.1908 moles of C
C
Equilibrium Constant is
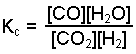
Since Kc is being determined, check to see if the given equilibrium amounts are expressed in moles per liter (molarity). In this example they are not; conversion of each is requried.
[C
[
[CO] = 0.0046 M
[

