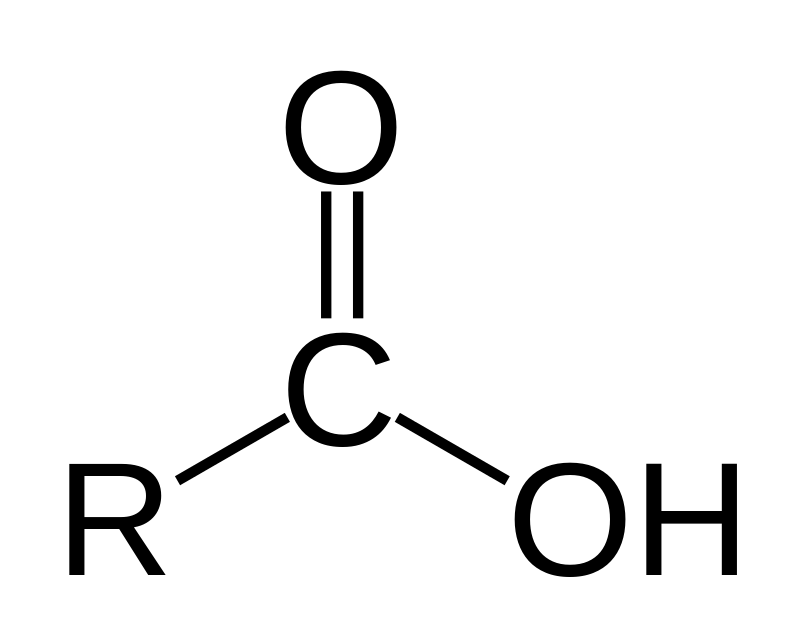
What is the general formula of a carboxylic acid?
1 Answer
Nov 15, 2014
A carboxylic acid has the formula
There are five different compounds with this general formula, where the only change is what is bonded to the carbon at the 135 degree angle, and which R group is bonded at the 225 degree angle. These compounds are: aldehyde, carboxylic acid, ketone, ester and amide.